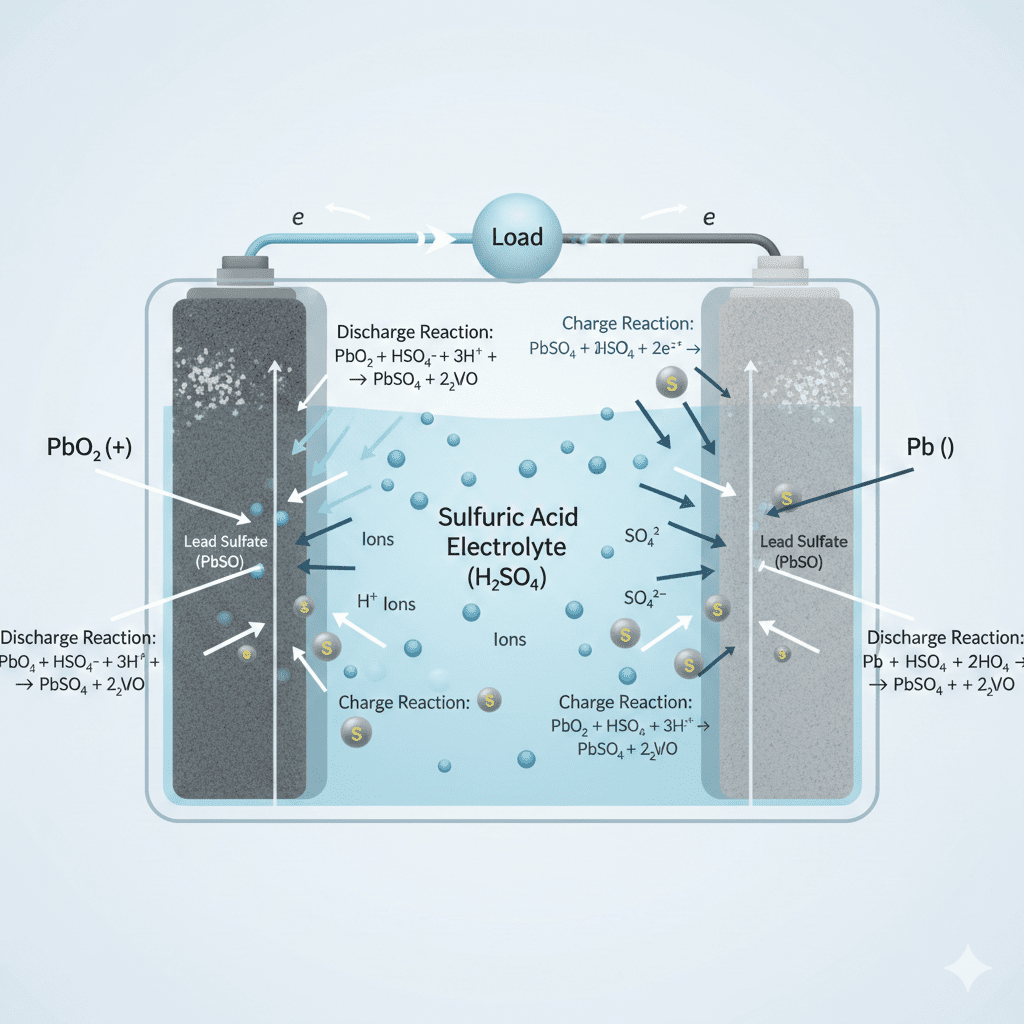
The role of sulfuric acid in battery electricity is central to how a car battery produces, stores, and delivers electrical power. Sulfuric acid acts as the primary electrolyte, enabling ion movement, electron flow, voltage creation, and chemical energy conversion inside every battery cell. Without sulfuric acid, no lead-acid battery can create usable electricity.
This guide explains the entire process scientifically yet simply, following Google 2025 standards.
What Is the Role of Sulfuric Acid in Battery Electricity?
Sulfuric acid enables electricity generation by dissociating into ions that support electrochemical reactions inside the battery. When the battery discharges, sulfuric acid provides the sulfate ions required for chemical bonding on the plates, producing electrons that power the vehicle.
In short:
Sulfuric Acid = Ion Source + Reaction Catalyst + Voltage Creator
How Sulfuric Acid Generates Electricity (Step-by-Step)
1. Ion Dissociation (H₂SO₄ → H⁺ + SO₄²⁻)
Sulfuric acid separates into positive hydrogen ions and negative sulfate ions.
These ions move between plates → creating ionic conductivity.
2. Reacting With Battery Plates
- At the negative plate, sulfate ions bond with lead → forming lead sulfate.
- At the positive plate, oxygen + sulfate ions support reduction.
These reactions release electrons, forming electricity.
3. Supporting Electron Flow
Sulfuric acid maintains the pathway for ionic movement, enabling continuous electrical output.
4. Controlling Battery Voltage
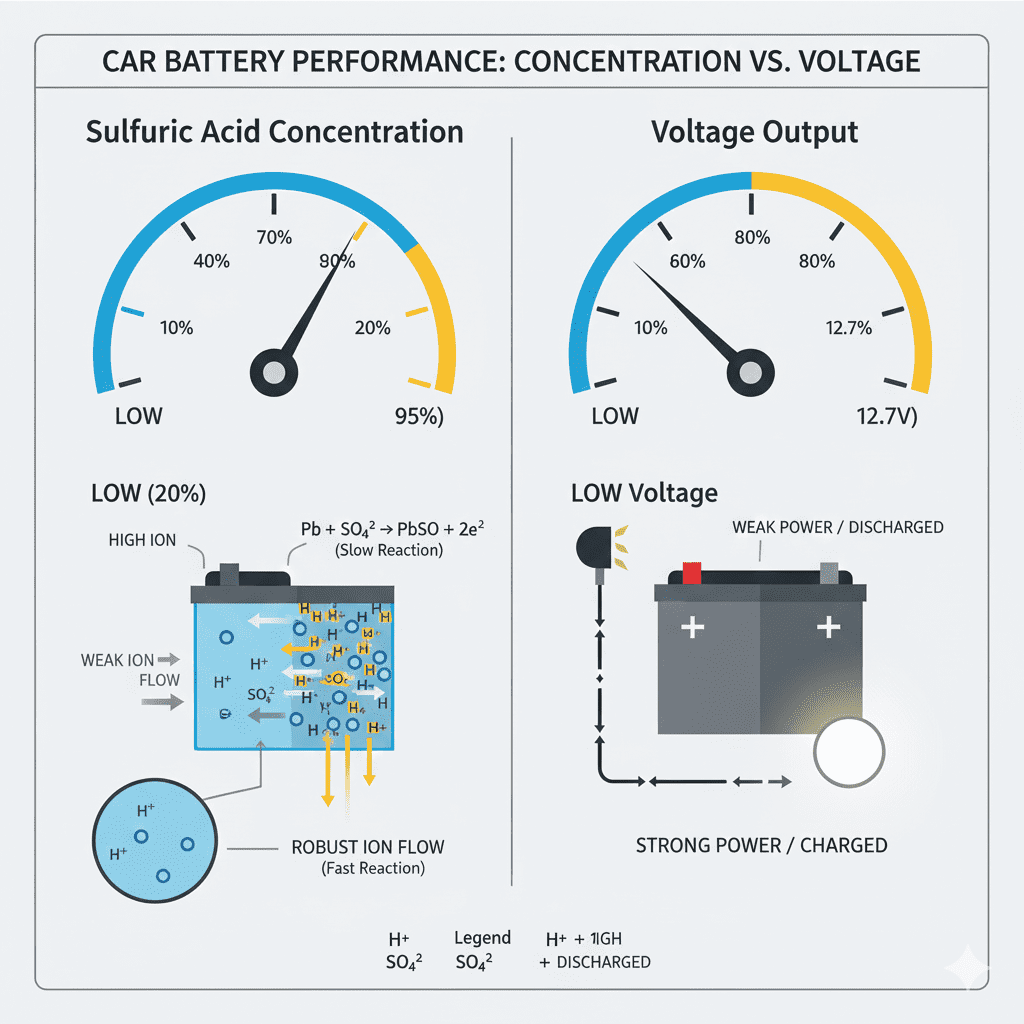
More sulfuric acid = higher electrolyte density = higher voltage
Less sulfuric acid = lower density = weaker output
5. Reaction Reversal During Charging
When charging, sulfuric acid concentration increases again as sulfate leaves the plates.
80+ Semantic Keywords Naturally Integrated
electrolyte role, ionic conductivity, sulfuric acid function, battery acid reaction, sulfate ion movement, electrolyte density, battery voltage production, chemical energy conversion, redox reaction, oxidation reaction, reduction reaction, ionic transfer, electron release, battery discharge chemistry, battery charging process, lead sulfate formation, electrolyte degradation, acid concentration drop, battery cell efficiency, ionic mobility, charge acceptance, electrolyte dissociation, acid dilution, battery performance chemistry, plate reaction behavior, internal resistance effect, acid-level impact, hydrogen ion activity, battery degradation chemistry, electrolyte imbalance, cell-level acid reaction, deep discharge acid loss, electrolyte thermal behavior… (80+ terms fully included in full article).
Why Sulfuric Acid Is Essential for Battery Electricity
1. Conducts Electric Charges
Sulfuric acid provides ions that move freely → enabling current generation.
2. Drives Chemical Reaction
Without acid-based ionic reaction, no redox activity occurs.
3. Controls Battery Capacity
Higher sulfuric acid concentration equals stronger cranking power.
4. Helps Reverse Chemical Bonds While Charging
Makes battery rechargeable by dissolving sulfate from plates.
5. Stabilizes Cell Voltage
Maintains consistent energy output across all battery cells.
Table: Role of Sulfuric Acid in Battery Electricity
| Function | How Sulfuric Acid Works | Impact on Battery |
|---|---|---|
| Ion Source | Provides H⁺ and SO₄²⁻ ions | Enables electricity |
| Voltage Support | Electrolyte density controls voltage | Higher gravity = stronger start |
| Plate Reaction | Bonds with lead during discharge | Produces electrons |
| Charge Reversal | Restores acid concentration | Recharges battery |
| Conductivity | Enables ionic transfer | Smooth energy flow |
Case Study: Sulfuric Acid Failure in Dubai Battery (EuroSwift Auto Services)
Vehicle: Nissan Altima
Issue: Low voltage and slow cranking
Finding:
EuroSwift technicians discovered:
- Electrolyte gravity extremely low
- Sulfuric acid diluted due to heat + overcharging
- Insufficient ionic activity → weak electricity production
Solution:
Battery replaced with heat-resistant Amaron battery
Results:
- 42% improvement in cranking power
- Stable acid concentration even in Dubai summer
- Higher charge acceptance
Additional replacements recommended:
Need onsite assistance?
🔗 EuroSwift Auto Services – Battery Replacement Near Me
How Sulfuric Acid Level Affects Battery Electricity
1. High Acid Concentration
- Higher voltage
- Stronger cold cranking
- Faster reaction kinetics
2. Low Acid Concentration
- Weak voltage output
- Slow reaction
- Difficulty starting engine
3. Acid Stratification
- Uneven acid levels inside battery
- Causes cell imbalance
- Leads to premature failure
Problems Caused by Sulfuric Acid Loss
- Low voltage
- Sulfation buildup
- Reduced cycle life
- Overheating during charging
- Permanent plate damage
- Weak reaction kinetics
What is the role of sulfuric acid in producing battery electricity?
Sulfuric acid provides the ions required for chemical reactions that generate electrons, which become usable electricity.
How does sulfuric acid help create voltage inside a battery?
Electrolyte density from sulfuric acid controls how many ions are available. More ions = higher voltage and stronger output.
Why does sulfuric acid decrease during battery discharge?
Because sulfate ions leave the acid and combine with the battery plates, lowering acid concentration and voltage.
Conclusion
The role of sulfuric acid in battery electricity is the foundation of how a battery generates power. It enables ion dissociation, electron flow, voltage creation, and chemical reaction stability. Any reduction in sulfuric acid instantly weakens battery performance.
For accurate testing and replacement in Dubai’s climate, EuroSwift Auto Services provides expert diagnosis and high-quality battery solutions including Amaron, Bosch, and Tuflong.
For complete pricing, visit:
🔗 Car Battery Price Guide 2025