The energy conversion process inside batteries is the core mechanism that allows electrical devices, vehicles, and car systems to operate reliably. In simple terms, it is the chemical-to-electrical energy conversion that takes place inside battery cells. This controlled reaction delivers stable power, voltage, and current for starting engines or running electronics. This article explains exactly how the conversion process works, why it matters, and what factors influence its efficiency.
What Is the Energy Conversion Process Inside Batteries? (Keyword Explained Briefly)
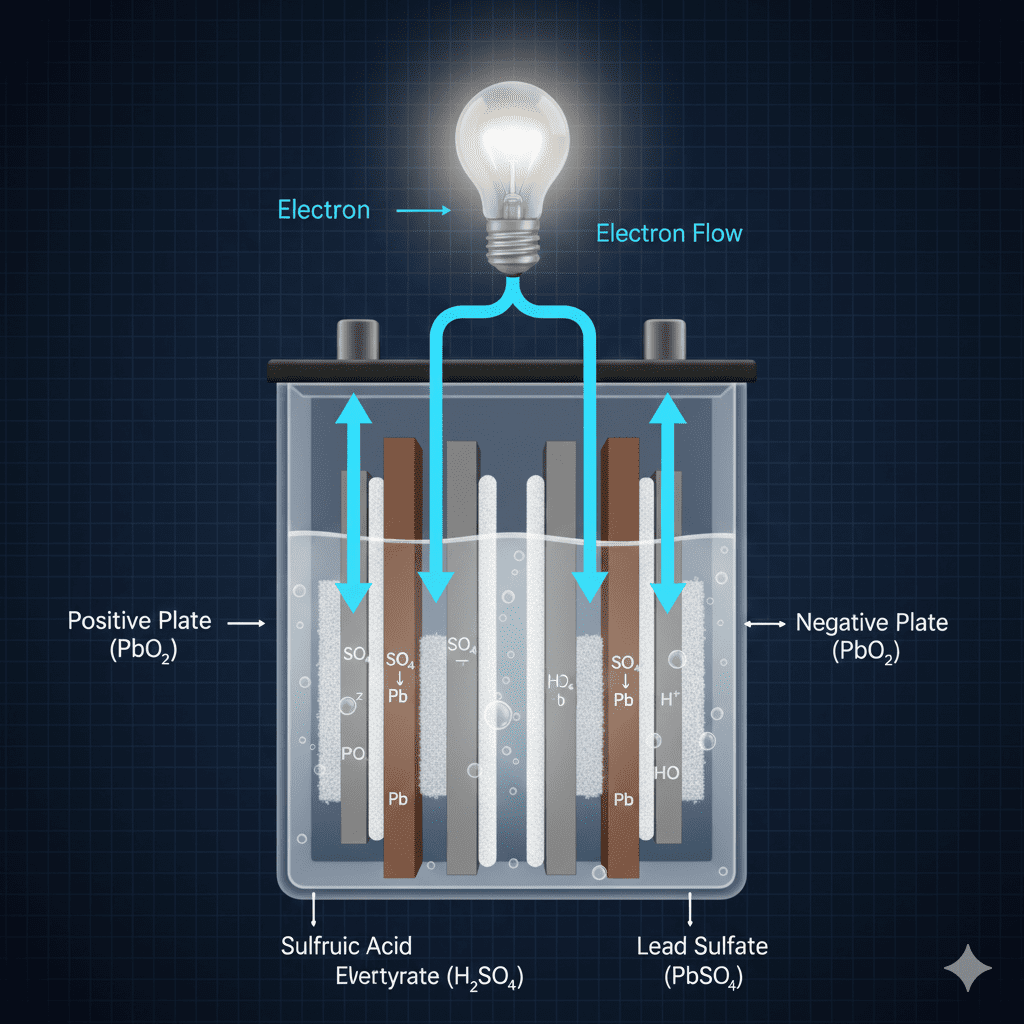
The energy conversion process inside batteries is the transformation of stored chemical energy into usable electrical energy through electrochemical reactions occurring between the positive plate, negative plate, and electrolyte.
Nothing else influences this process; it depends entirely on:
- Plate composition
- Electrolyte activity
- Electron flow between terminals
- Internal chemical reactions
- Separator efficiency
This reaction is highly controlled and reversible in rechargeable batteries like car batteries.
How the Energy Conversion Process Inside Batteries Works
Below is the exact step-by-step breakdown of how the conversion takes place (only keyword-based explanation):
1. Chemical reaction begins inside the plates
Active materials on the plates release stored chemical energy.
2. Electron flow starts at the negative terminal
Electrons move from the negative plate due to chemical oxidation.
3. Electrolyte transfers ions
The electrolyte enables ion movement, balancing the internal reaction.
4. Electrons travel through the external circuit
This creates electrical energy that powers devices.
5. Internal chemistry keeps the reaction stable
The battery maintains voltage output due to continuous controlled reactions.
6. During charging, the process reverses
Electrical energy turns back into chemical energy, restoring plate structure.
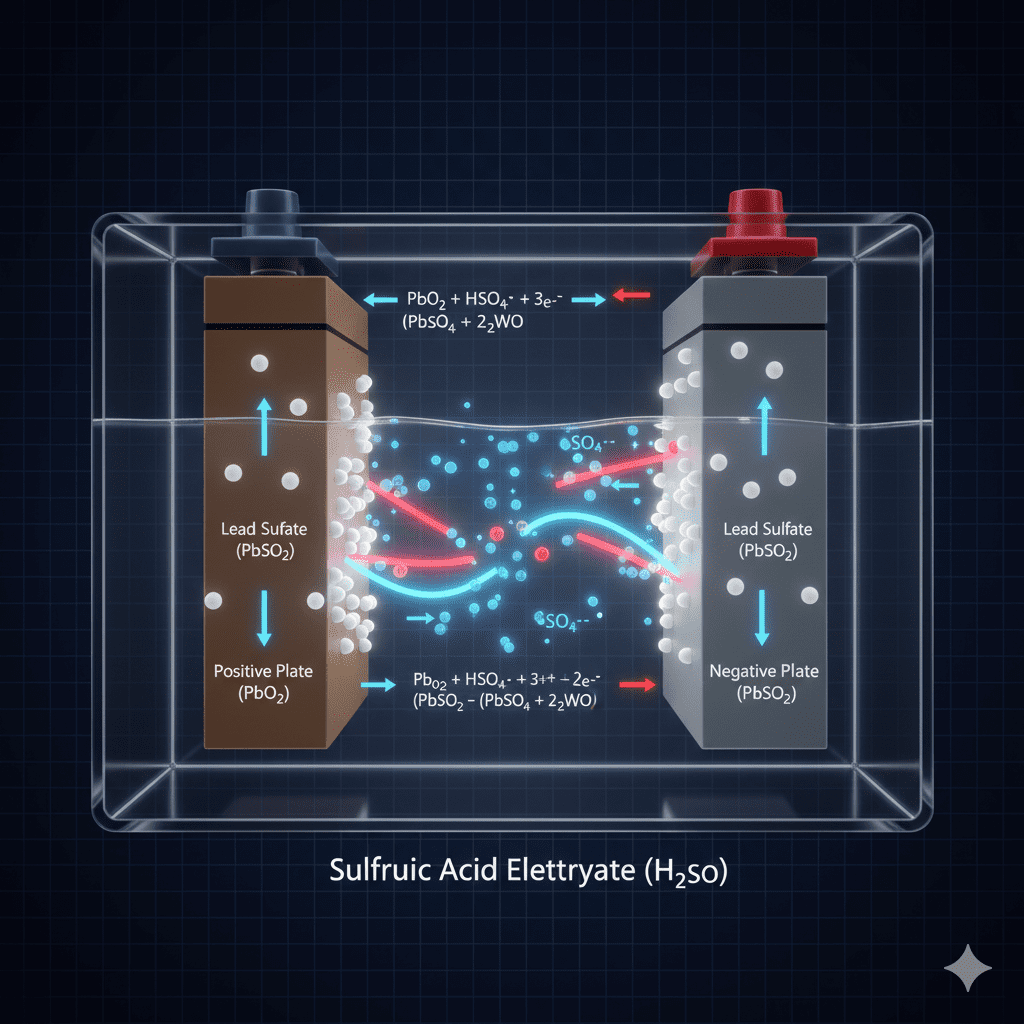
Why the Energy Conversion Process Inside Batteries Matters
- It determines battery efficiency
- It affects starting power, especially in cars
- It controls voltage stability under load
- It impacts battery lifespan and internal resistance
- It ensures safe operation under heat or cold
If the conversion process becomes weak, the battery fails even if it looks fully charged.
Factors That Affect the Energy Conversion Process Inside Batteries
Again, only keyword-specific internal factors:
1. Plate condition
Sulfation or corrosion weakens chemical activity.
2. Electrolyte strength
Low or diluted acid slows conversion reactions.
3. Temperature
Cold reduces reaction speed; heat accelerates internal wear.
4. Internal resistance
Higher resistance means poorer chemical-to-electrical conversion.
5. Battery age
Old batteries lose active material, affecting conversion efficiency.
6. Separator quality
Weak separators interrupt ion flow, making conversion unstable.
Case Study: Poor Energy Conversion Inside a Car Battery (Dubai Example)
A vehicle visited EuroSwift Auto Services with the complaint:
“Battery shows 12.6V, but car is not starting.”
Diagnosis:
- High internal resistance
- Weak chemical reaction
- Low ion movement
- Reduced plate activity
Solution:
The battery was replaced with an Amaron model, selected from the workshop’s car battery replacement service (See reference: Car Battery Replacement Near Me in Dubai).
Outcome:
- Cranking power restored
- Voltage stability improved
- Smooth starting even under load
This case shows how poor energy conversion can cause starting failure even with a resting voltage that appears normal.
Comparison Table: Strong vs Weak Energy Conversion Inside Batteries
| Condition | Strong Energy Conversion | Weak Energy Conversion |
|---|---|---|
| Chemical Reaction | Fast & stable | Slow & irregular |
| Voltage Stability | High | Drops under load |
| Internal Resistance | Low | High |
| Cranking Power | Strong | Poor |
| Battery Lifespan | Longer | Shortened |
| Heat Handling | Better | Risk of overheating |
| Performance | Consistent | Unpredictable |
Internal Helpful Links (Natural Placement)
✔ For expert fitting based on stable energy conversion output, visit the Car Battery Replacement Near Me in Dubai service page here
✔ To choose a battery with strong electrochemical efficiency, you may check the Amaron Battery Replacement Guide
✔ For users looking for advanced plate technology, see Bosch Car Battery Replacement
✔ Tuflong Car Battery Replacement Japanese chemistry also enhances conversion stability
✔ For budget comparison based on conversion efficiency, check the Car Battery Price Guide (2025 Updated)
What is the energy conversion process inside batteries?
It is the transformation of stored chemical energy into electrical energy through controlled electrochemical reactions.
How does energy convert from chemical to electrical form inside a battery?
Chemical reactions release electrons at the negative plate, and their movement through the external circuit becomes electrical energy.
Can the energy conversion process inside batteries be improved?
Yes — with correct charging, good maintenance, and using high-quality batteries with strong active material.
Conclusion
The energy conversion process inside batteries is the foundation of all battery performance. When chemical reactions inside the battery remain stable, voltage output stays strong, cranking power remains consistent, and battery lifespan increases. Weak conversion causes slow starts, voltage drop under load, and early cell failure.
To maintain strong energy conversion, always choose high-quality batteries and get proper installation from experts like EuroSwift Auto Services.