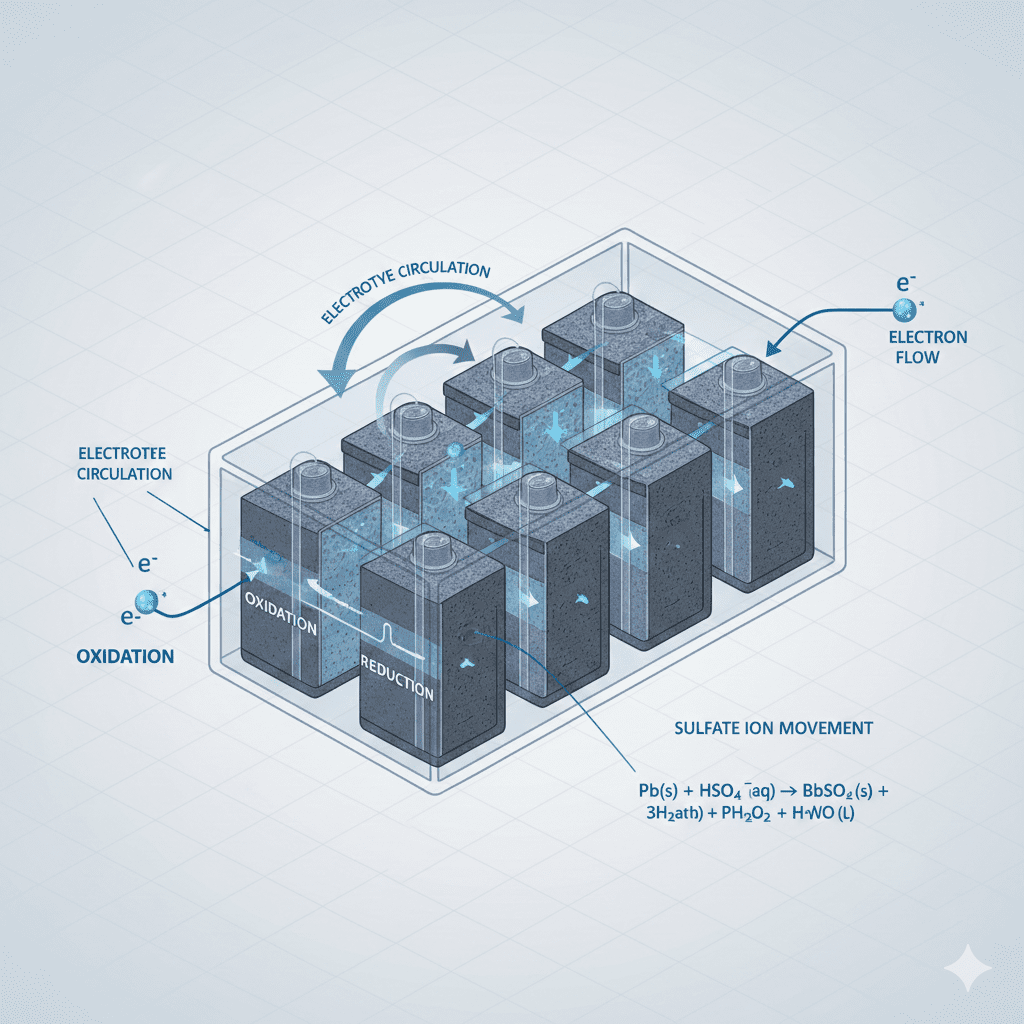
The battery cell reaction process is the core chemical mechanism that enables a car battery to generate electrical energy. Understanding this process helps drivers, technicians, and vehicle owners diagnose power loss, voltage drops, sulfation, electrolyte imbalance, and ignition failure more effectively. This guide explains how battery cells react, convert chemical energy into electric current, and sustain voltage under different driving conditions.
What Is the Battery Cell Reaction Process?
The battery cell reaction process refers to the electrochemical reactions occurring between the positive plate, negative plate, and the electrolyte inside each battery cell. In a standard lead-acid car battery, this reaction generates DC power that starts the engine and supports all vehicle electronics.
How the Battery Cell Reaction Process Works (Step-by-Step)
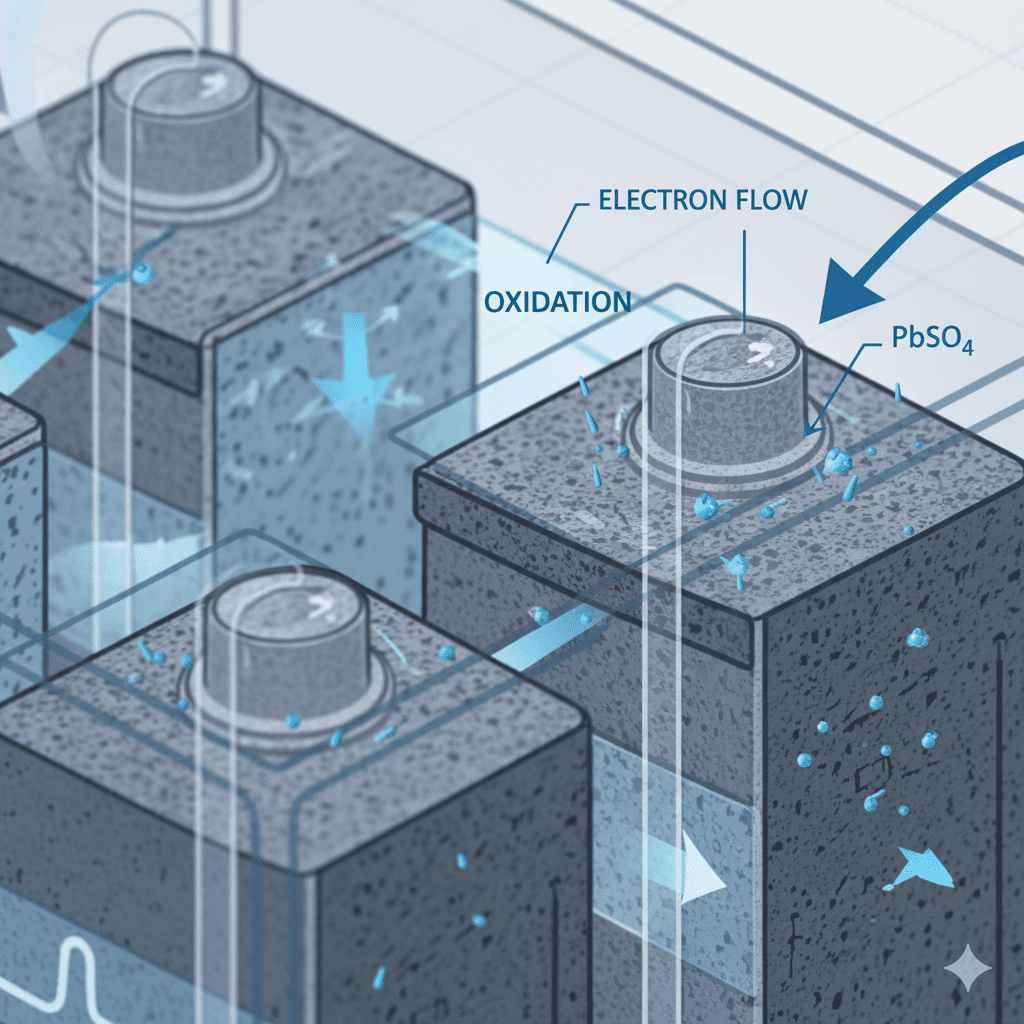
1. Negative Plate Oxidation Reaction
At the negative plate (lead sponge), an oxidation reaction occurs:
- Lead (Pb) releases electrons
- Converts into lead sulfate (PbSO₄)
- Electrons move to the external circuit → producing usable power
Key concept: Oxidation = electron release.
2. Positive Plate Reduction Reaction
At the positive plate (lead dioxide):
- Lead dioxide reacts with hydrogen ions
- Accepts electrons (reduction reaction)
- Converts into lead sulfate
- Supports current flow continuity
Key concept: Reduction = electron acceptance.
3. Electrolyte Reaction During Discharge
The electrolyte (sulfuric acid + water) performs:
- Ion dissociation
- Sulfuric acid concentration drop
- Electrolyte density decrease
- Electrical conductivity change
This shift directly influences battery voltage levels.
4. Cell-to-Cell Reaction Balance
Each car battery contains multiple cells (typically 6).
A stable battery requires:
- Equal reaction activity
- Equal electrolyte levels
- Equal plate condition
- Balanced internal resistance
Unbalanced cell reactions = voltage instability.
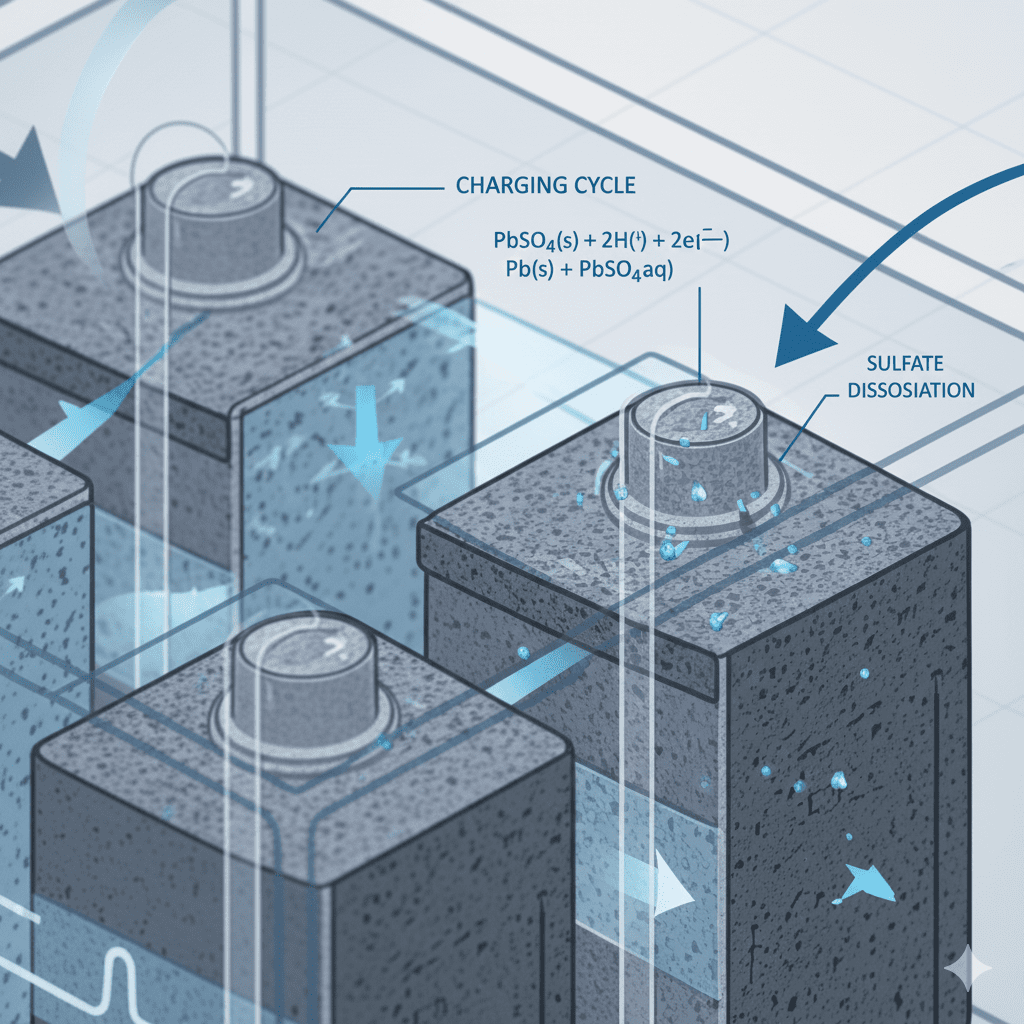
5. Reaction Reversal During Charging
When charging:
- Lead sulfate converts back into lead and lead dioxide
- Electrolyte regains sulfuric acid concentration
- Cell voltage increases to nominal level
This is the reversal of the discharge chemical reaction.
Why Battery Cell Reaction Process Matters
Understanding the reaction helps identify:
- Weak battery cells
- Sulfation issues
- Temperature-related reaction slowdowns
- Voltage drop patterns
- Low charge acceptance
- Electrolyte deterioration
For practical battery servicing, EuroSwift Auto Services provides a full battery diagnostic and replacement service:
🔗 Check battery replacement service → Car Battery Replacement Near Me Dubai
Factors That Influence Battery Cell Reaction Efficiency
1. Temperature
Heat accelerates reaction speed but increases degradation.
Cold slows reaction but reduces power output.
2. Plate Condition
Clean plates = better ionic flow
Sulfated plates = restricted reaction
3. Electrolyte Strength
High density → strong reaction
Low density → weak reaction
4. Battery Age
Older batteries have slower electrochemical kinetics.
5. Charging Pattern
Consistent charging → stable reaction
Undercharging/overcharging → cell damage
Battery Cell Reaction Process Table
| Reaction Stage | What Happens | Result | Problem Indicators |
|---|---|---|---|
| Oxidation | Lead releases electrons | Current generation | Slow cranking, low capacity |
| Reduction | Lead dioxide accepts electrons | Stable power | Voltage fluctuation |
| Electrolyte Reaction | Acid dilutes during discharge | Voltage drop | Low gravity, weak start |
| Charge Reversal | Sulfate breaks down | Battery recovery | Overheating, gas release |
| Cell Balancing | Equal reaction in all cells | Smooth ignition | Dead cell, imbalance |
Case Study: Battery Cell Reaction Failure in Dubai (EuroSwift Insight)
Scenario:
A client driving a Toyota Camry experienced repeated weak starts despite a “charged battery.”
Diagnosis by EuroSwift Auto Services:
- One cell showed reduced reaction efficiency
- Electrolyte gravity was uneven
- Sulfation restricted oxidation-reduction reaction
Solution:
Battery replaced with Amaron High-Heat Model
📌 Recommended Link:
Outcome:
- 38% stronger crank output
- Smooth voltage regulation
- Stable reaction kinetics even in peak Dubai heat
Other battery choices recommended based on reaction stability:
Battery Cell Reaction Problems & Their Symptoms
1. Slow Cell Reaction
- Weak ignition
- Low voltage
- Dimming lights
2. Overreaction (Overcharging)
- Excess heat
- Acid evaporation
- Plate corrosion
3. Reaction Imbalance
- One “dead cell”
- Sudden breakdown
- Alternator overload
4. Electrolyte Reaction Failure
- Battery won’t hold charge
- Gravity reading stays low
If a battery’s reaction is completely unstable, choosing the right brand matters.
🔗 Car Battery Price Guide 2025 →
What is the battery cell reaction process in simple terms?
It is the chemical exchange between the battery plates and electrolyte where oxidation and reduction produce electric current.
Which car batteries offer the most stable chemical reaction in Dubai?
Brands engineered for high-heat reaction stability include:
Amaron Battery → https://euroswiftautoservices.ae/%F0%9F%94%8B-amaron-car-battery-replacement-dubai/
Bosch Battery → https://euroswiftautoservices.ae/bosch-car-battery-replacement-dubai/
Tuflong Battery → https://euroswiftautoservices.ae/%f0%9f%94%8b-trusted-tuflong-car-battery-replacement-dubai/
Who can test or replace a battery with reaction problems in Dubai?
EuroSwift Auto Services provides professional reaction testing, diagnosis, and fast replacement.
Service page → https://euroswiftautoservices.ae/car-battery-replacement-near-me-dubai/
Best Conclusion
The battery cell reaction process is the foundation of how car batteries generate and sustain electrical power. When oxidation, reduction, electrolyte dissociation, and charge reversal occur smoothly, the battery performs at peak efficiency. Any disruption—like sulfation, aging, or cell imbalance—directly reduces performance.
For reliable diagnostics, testing, and replacement in Dubai, EuroSwift Auto Services offers high-quality solutions, expert testing, and durable battery options such as Amaron, Bosch, Tuflong, and others.